What is particulate? How does it origin? Which problems does it create?
Particulate: formation, effects and treatment!
Before think the merry flames of a fireplace, then a dish that is cooking on a gas stove. Which types of images do you create in your mind? Probably, in the first case a restless yellow and red flame, which constantly changes shape. In the second case, however, it will probably emerge in your mind the image of a quiet blue flame. What is the difference between the two?
In the course of the article we will provide answer to this question by analyzing one of the best known forms of pollution: the atmospheric particulate.

In particular, we will analyze together three different aspects which characterize the particulate.
- What is it?
- How does it form?
- What effect has on the environment and health?
Let’s start by answering the first of these three questions. The information contained in this article are mainly taken from an interesting analysis written on Chimica & Ambiente magazine [1].
What is the particulate?
Is it called the set of particulate air pollution in the atmosphere, coming from natural or anthropogenic source. In fact the earth’s atmosphere, whether you analyze urban areas is that you analyze remote areas, has a high concentration of particles (of the order of 108 to cm3). It is not so much the number of particles that counts, since their feature size.
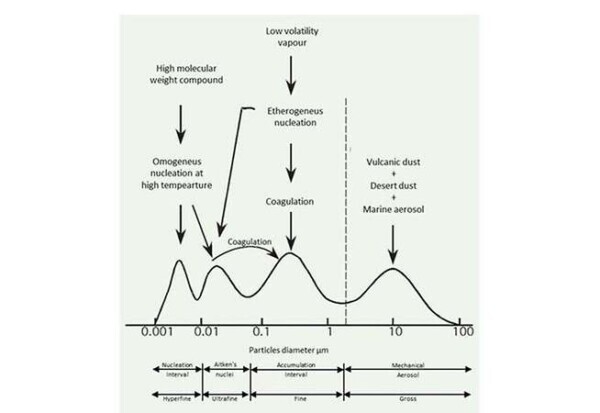
In this interesting image, taken from the article mentioned above [1], we can see how the size of the various particles cover a very wide range, ranging from nanometer-size to hundreds of microns. To make a discussion as thoroughly as possible of particulate air pollution, it appears to be useful to consider different ranges of size characteristics:
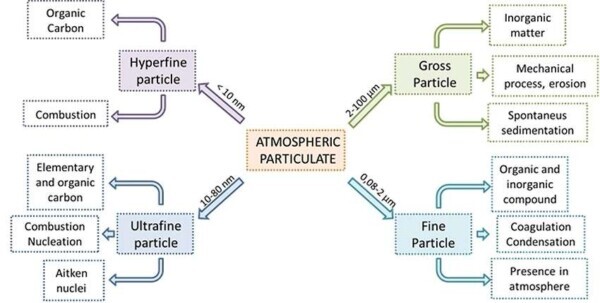
- 2-100 µM: This interval corresponds to the coarse particulates, and typically comes from mechanical processes and erosion, marine aerosols and desert sand. These particles are mainly of inorganic, highly dependent on the geographical area in which they are taken, and also include biogenic compounds (pollen, spores, …). It is important to emphasize that for particles of this size, the gravitational contribution is more significant than the motion of thermal agitation, and it is therefore marked their tendency to spontaneous sedimentation. The characteristic time of permanence in the atmosphere turns out to be small.
- 0.08 to 2 µM: This interval is known as an ‘accumulation’, and is characterized by fine particles. The particles are characterized by these dimensions are typically formed by coagulation of smaller particles or for condensation of chemical species of pre-existing particles. Given the small size, it is had that the energies associated to the gravitational action and Brownian motion are of similar orders of magnitude, resulting in low tendency to sedimentation. The fine particulate matter tends to remain in the atmosphere, and until it is removed from the rain in a profound way influence the chemical equilibrium of the atmosphere itself.
- 10-80 nm: The particles that belong to this size range are also referred to Aitken nuclei and form the ultrafine particles. They can be formed by the nucleation mechanisms of supersaturated vapors at low temperature, or within the context of the combustion processes. It is precisely these particles, by means of condensation phenomena, increase their size and contribute to the creation interval of accumulation. They consist almost entirely of elemental or organic carbon.
- <10 nm: A series of new experimental techniques allows to analyze increasingly smaller orders of magnitude, and go to below 10 nm. Particles smaller than this numerical value, consisting mainly of organic carbon, form the hyperfine particles and they are in the interval defined nucleation.
Now that we have examined what the particulates and various geometrical dimensions that characterize it, is appropriate to carry out in depth its origin. In particular, we will examine how the particles is produced in combustion processes.
How does particulate is form?
If the processes of formation of many pollutants are clear, the same can not be said for the formation of the particulates. The steps that lead to the formation of these carbonaceous particles are very complex, and are characterized by intricate reactions between the multiple chemical species that are generated in combustion processes.
In the article dedicated to the TAR, where we analyzed the definition of equivalent ratio and the methane combustion mechanisms. In that same article we had seen as the onset of high concentration of fuel zones (Φ >> 1) leads to the development of pyrolytic conditions, in which the termination of two methyl radicals and subsequent dehydrogenation give rise to the formation of acetylene. Acetylene, in turn, allows the genesis of benzene and species of poly-aromatic (PAH) compounds. It is from these aromatic species that form soot and black carbon!
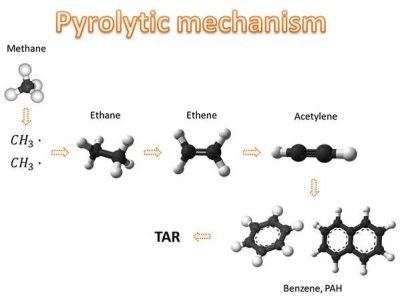
The poly-aromatic compounds have a planar geometry, but their growth induces Van der Waals forces to operate in coagulation processes, with consequent assumption of three-dimensional geometry. In this way they generate the primary soot particles, which subsequently undergo graphitisation processes and surface growth. From these particles, by means of coagulation mechanisms and absorption, the structures are generated with larger diameter characteristics of fine and ultrafine particles. If these structures were to meet some rich areas of combustion, we have that the characteristic times of oxidation of polyaromatic compounds and carbon particles are such as to make them difficult to complete oxidation. In other words, they will meet such structures in the outgoing gas flow. These considerations bring out an important warning:avoid the creation of fuel-rich zones in combustion processes!
So, how to promote adequate mixing?
In the gas phase there are two different modes of management of the combustion process:
- Premixed Flames: In this case, fuel and oxidizing agent are mixed at a low temperature. The perfectly homogeneous mixture is then sent to the combustion chamber: on the one hand there are the flame formed by fuel and oxidizing agent, while the on the other side of the process there are products of oxidation. Although this operating mode introduces the considerable advantage of not forming particulate, the other side of the coin can present safety issues. A trigger source can lead power of the mixture even outside the combustor, and will therefore seek to complete the mixing process in the vicinity of the flame. This is how it is managed combustion in stoves: the blue color is characteristic of flames with little soot.
- Diffusive Flames: n Flames diffusive fuel and the oxidant are fed separately, and are mixed using transport material phenomena laminar or turbulent. Since the reagents are mixed in the vicinity of the flame front, it is likely that any inefficiencies lead to the formation of fuel-rich zones, resulting istaurazione of pyrolytic mechanisms and particle formation. The particulate, the temperatures of the flames characteristics, has an emission spectrum that gives the same flames the characteristic yellow-orange color. Therefore, although this mode of operation is generally more secure, it has the disadvantage of promoting increased formation of pollutants species.
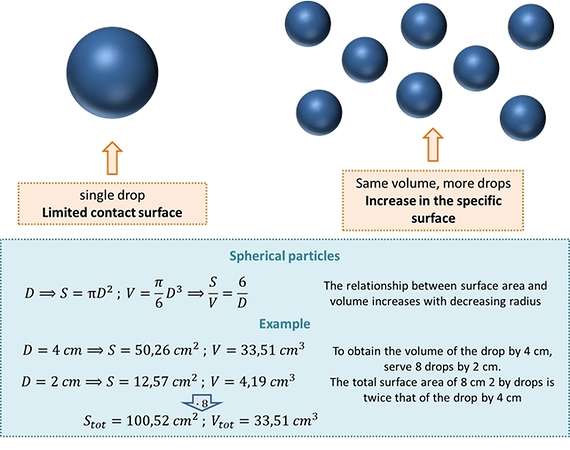
If we consider the combustion of liquid and solid species, we can notice that the mixing phenomena are further complicated by the presence of different phases. In this case the key to better manage the oxidative process is to ensure a large contact area between the fuel (solid or liquid) and the comburent (gaseous). In the case of liquids, this is achieved by creating a fine dispersion of droplets. In the case of solids, could be useful to try to use material with a reduced feature size.
Now that we have analyzed the nature of particles and the mechanisms that generate it, we make considerations about the consequences that this pollutant has for the environment and to humans.
Which type of effects has got on health and on enviroment?
Imagine to clean a yard full of small rocks, and then clean a sand-filled courtyard. The two series are very different: the size characteristics of the rocks are significantly higher than those of sand, and this makes them easier to remove.
Even for the health and the environment, the impact appears to be closely related to the size characteristics of the particles involved. We begin by examining the health effects.
Health effects
The health effects of particulate matter are significant. From epidemiological studies it has shown that there is a correlation between the presence of particulates in the atmosphere, and an increase of the levels of mortality and hospitalization. Consider that in London, in 1952, they were recorded mortality increases on the order of 4000 deaths a week to a concentration of particles in the order of 4 mg / m3.
Today, fortunately, thanks to technology development of production processes, the concentration of dust in the air has been drastically reduced. It is also true that the current pollution levels are not harmless: nevertheless have consequences for health, and in particular, some studies have shown that the most significant consequences were related to smaller particles. The size of the particles are the same, therefore, the main parameter to consider its harmfulness to human health. The cells in fact react with the surface of the particles, rather than with their volume: the presence of adsorbed materials on the surface can then depositing harmful compounds in the same cells.
As you can imagine, the apparatus hardest hit by this category of pollutants is the breathing. The size of the particles considerably influence their degree of penetration into the respiratory tract.

In particular, the particles having a size greater than 1 micron are easily intercepted and deposited in the nose and throat, and then be duly expelled. The particles between 100 nm and 1 micron can reach up to the bronchioles, there they are conveyed to the throat and be removed in a relatively short time. If we consider of still smaller size, the situation becomes more problematic, since the particles are not retained by the nasal or bronchial mucosa and can penetrate to the alveoli. In this case the removal mechanisms appear to be less efficient and less complete, with important consequences for health.
If we consider the hyperfine particles, it could emerge at the neuronal level of problems.
A study by the University of Rochester [2] has indeed shown that the hyperfine particles can interact directly with the central nervous system, settling near the olfactory nerve endings and spreading to the brain. Taking into account the potential accumulation of these particles, they may emerge of severe neurotoxic effects.
Environmental effects
The environmental effects of particulate matter are quite complex. In fact aerosols dispersed into the atmosphere are involved in a complex manner with the thermal radiation incident and reflected from the Earth, as a function of their concentration, morphology, optical properties and solubility. We can distinguish different aerosol families, and separately analyze their effects on the Earth’s climate.
- Sulfates: They are the most studied aerosols and show no absorption bands in the visible range. It is therefore considered that give a negative contribution to the increase of the solar radiative flux, since scatter the solar radiation that index on the earth’s surface
- Elementary Carbon: It has a very broad absorption spectrum, ranging from ultraviolet to infrared. This substance can therefore contribute both negatively (by dispersing the incoming solar radiation) that positively (by absorbing infrared radiation emitted from the earth) to radiative forcing. Estimates quantify its total contribution as positive
- Organic Carbon: this type of compounds present in the sample of the problems is that in the analysis, and thus do not have a clear idea about its influence on climate

In addition to the direct contribution of aerosols it is of fundamental importance to analyze their indirect contribution. The particles in fact facilitate the genesis of droplets in clouds and fogs, acting as nuclei on which condensation takes place of water molecules. In high pollution areas it is had that the particles contribute greatly to the number of drops of water that make up the clouds, contributing to reflection of the incident solar radiation.
Thus, we have seen many on particulate information, how it is formed and which influence has got on health and environment. In a new article we want to answer a key question: how is it possible to remove particulates that is formed in various industries?
In the meantime, look at our Case Study and discover our solutions to reduce pollutants!
See you soon with new information!
Bibliography
[1] A. D’Alessio, A. D’Anna, T. Faravelli e E. Ranzi, «Particolato fine e ultrafine,» Chimica & Ambiente, pp. 34-42, 2005.
[2] G. Oberdörster, Z. Sharp, V. Atudorei, A. Elder, R. Gelein, W. Kreyling e C. Cox, «Translocation of Inhaled Ultrafine Particles to the Brain,» Inhalation Toxicology, pp. 437-445, 2004.