Neutralizing system for bag filters in fumes treatment
How does they work? Which benefits they imply? Check out our new article!
In the following article we talk about a very important practice in fumes treatment: the neutralization (inertization) of fumes. What does this process consist of ? When and why is it realized? Let’s find out together!
Inertization and fields of application
Inertization is a practice that is carried out in filtering plants for fumes treatment and it is realized to protect filtering elements from acid substances and unburnt particles. Usually, the product used in this process is calcium hydroxide (also known as slaked lime or hydrated lime), also called lime or quicklime, chemical formula Ca(OH)2. Lime results from dry hydration of calcium oxide. Hydrated lime looks like a white powder (lime powder) that, due to its chemical characteristics, is able to reduce the acidity present in the fumes (it is used also in agriculture and in other contexts).

Using lime we can:
- reduce the permeability of the filtering bags: the lime deposited in the interstices of the filtering surface prevents small particles from entering, thus increasing the efficiency of filtration;
- neutralize acid pollutants: thanks to its chemical characteristics, calcium hydroxide is suitable for the abatement of acid pollutants (HF, HCl and SO2) present inside the fumes to be purified. This feature is of fundamental importance as it also avoids the degradation of the filtering surface;
- protect the filter elements from any unburnt particles: we may find particles that have not completed the combustion process remaining dispersed in the air flow. These particles have a strong tendency to aggregate. Lime is able to avoid this behaviour and reduce the risk to damage filtering media (e.g. reduce fire risk).
This neutralizing process, therefore, led to a series of important advantages, but what are the fields of application?
As we have seen, it is used for the treatment of high temperature fumes that, for example, can be generated by biomass plants, thermoelectric power plants, incinerators and from the treatment of hazardous and non-hazardous waste.
Inertization: how does it work?
We have analyzed the general characteristics of this process (definition, product used and main advantages), but how does the inertization of fumes work? To understand this, we can analyze the following figure, which represents a typical fume treatment plant complete with inerting system with hydrated lime.
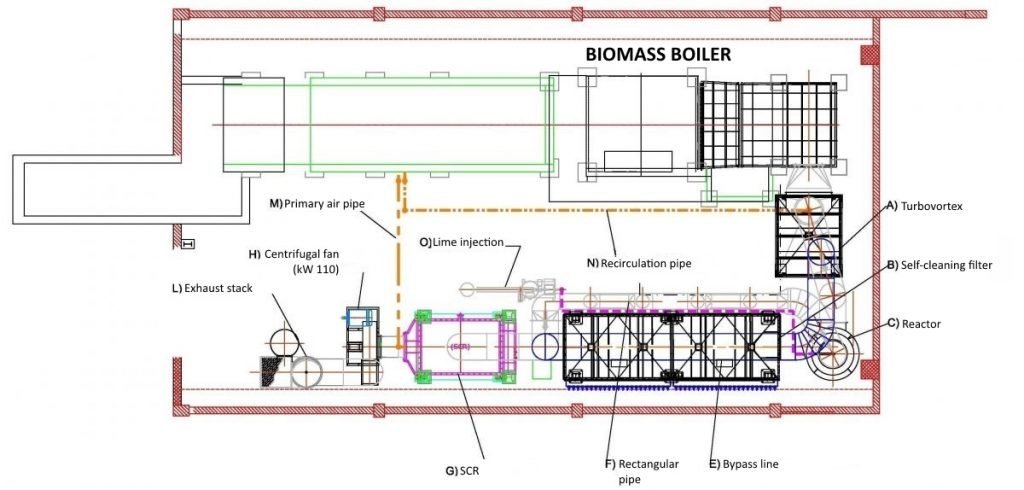
As we can see, the air flow is conveyed into the multi-cyclone Turbovortex® which is suitable as a pre-filter for dust present in the fumes.
This first step, however, may not be necessary for some types of plants as it is closely linked to the nature of the pollutants present: in case of high dust loads, the multi-cyclone allows to protect the main filtering unit(s) from excessive working conditions. After this first phase, the polluted flow passes through the reactor where fumes come into contact with calcium hydroxide. This step is very important because, in order to be effective on fume’s acidity, it is important to gain a very good mixing and an adequate contact time between the two elements.
As shown in the previous drawing, the reactor is connected to a storage silos which, by means of a dosing system, supplies the necessary quantity of lime. After fumes are homogeneously mixed with lime, the flow is conveyed to the bag filter where the particulate is treated.

Large systems, like the one in the figure, can also be equipped with other elements such as:
- pre-heating systems: used to heat up the filtering body in order to reach the most suitable temperature for correct operation and to avoid dew point. The choice to use or not this “add-on” is determined by the effective difference between internal and external thermal conditions, with the aim not to have any condensation inside the filter, so avoiding risk of clog for filtering bags and risk of corrosion to metal elements;
- insulation: this practice allows to thermally insulate filter surfaces avoiding heat dispersion. Depending on the operating temperatures, it can be made using stone wool (suitable for use at temperatures below 300 °C) or using ceramic materials (resistant even at very high temperatures);
- bypass line: applied to allow a full bypass of the filter in event of maintenance or until the fumes reaches proper temperature for filtration phase;
- SCR Unit: (Selective Catalytic Reduction) for the reduction of NOx which may be present in the fumes to be treated;
Smaller plants, usually, are not equipped with pre-heating systems so, during start-up phase, bags are covered with lime to reduce their permeability and filter is by-passed until fumes have reached target operating temperatures, usually higher than 150/160 °C and below 230 °C.
Filtering bags: materials and temperatures of use
When a bag filter is designed for high temperatures, it is very important to choose filtering media that are most suitable for the process type and operating temperatures. In the following table we show some details regarding filtering media for high temperatures:
| TYPE | WEIGHT | TEMPERATURE |
| Bag with polyphenylsulfide needle felt on polyphenylsulfide support | 550 g/m2 | Max 150° with peaks of 160° |
| Bag with polyphenylsulfide needle felt on PTFE support | 550 g/m2 | Max 160° with peaks of 180° |
| Bag in glass fibre with microporous membrane | 750 g/m2 | 250° with peaks of 260° |
| Bag with PTFE needle felt on PTFE support | 750 g/m2 | 250° with peaks of 260° |